Items on this page may include news associated with the IGNITE network as well as other organizations in the field of human genetics and genomics.
CYP2D6 phenotype and post-surgical pain control with hydrocodone and oxycodone
Christelle Lteif; Larisa H. Cavallari DOI: 10.3389/fphar.2026.1817652 Article on Jama Network...
ADOPT PGx Depression Manuscript Published
A new manuscript from the IGNITE Pragmatic Trials Network has been published in JAMA Network Open, describing results from the ADOPT PGx Depression trial. The randomized clinical trial evaluated whether […]...
Genotype-Guided Antidepressant Prescribing for Patients With Depression
Kathryn V. Blake, PharmD; Lindsay J. Hines, PhD; Michelle Liu, PharmD DOI: 10.1001/jamanetworkopen.2026.10609 Article on Jama Network...
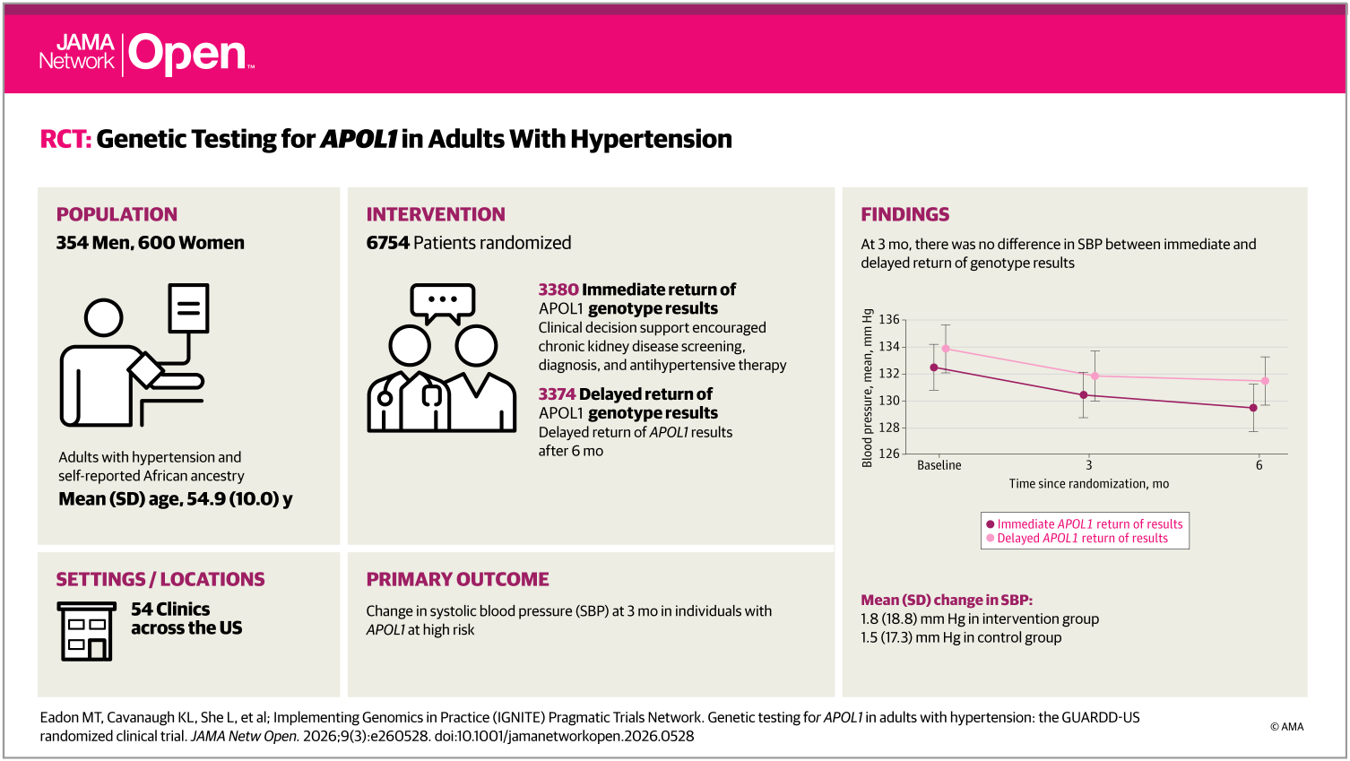
GUARDD-US APOL1 Genetic Testing Manuscript Published
A new manuscript from the IGNITE Pragmatic Trials Network has been published in JAMA Network Open, describing the primary results of the GUARDD-US study. GUARDD-US is a randomized controlled trial […]...
Genetic Testing for APOL1 in Adults With Hypertension – The GUARDD-US Randomized Clinical Trial
Michael T. Eadon, MD; Kerri L. Cavanaugh, MD, MHS; Lilin She, PhD DOI: 10.1001/jamanetworkopen.2026.0528 Article on Jama Network...
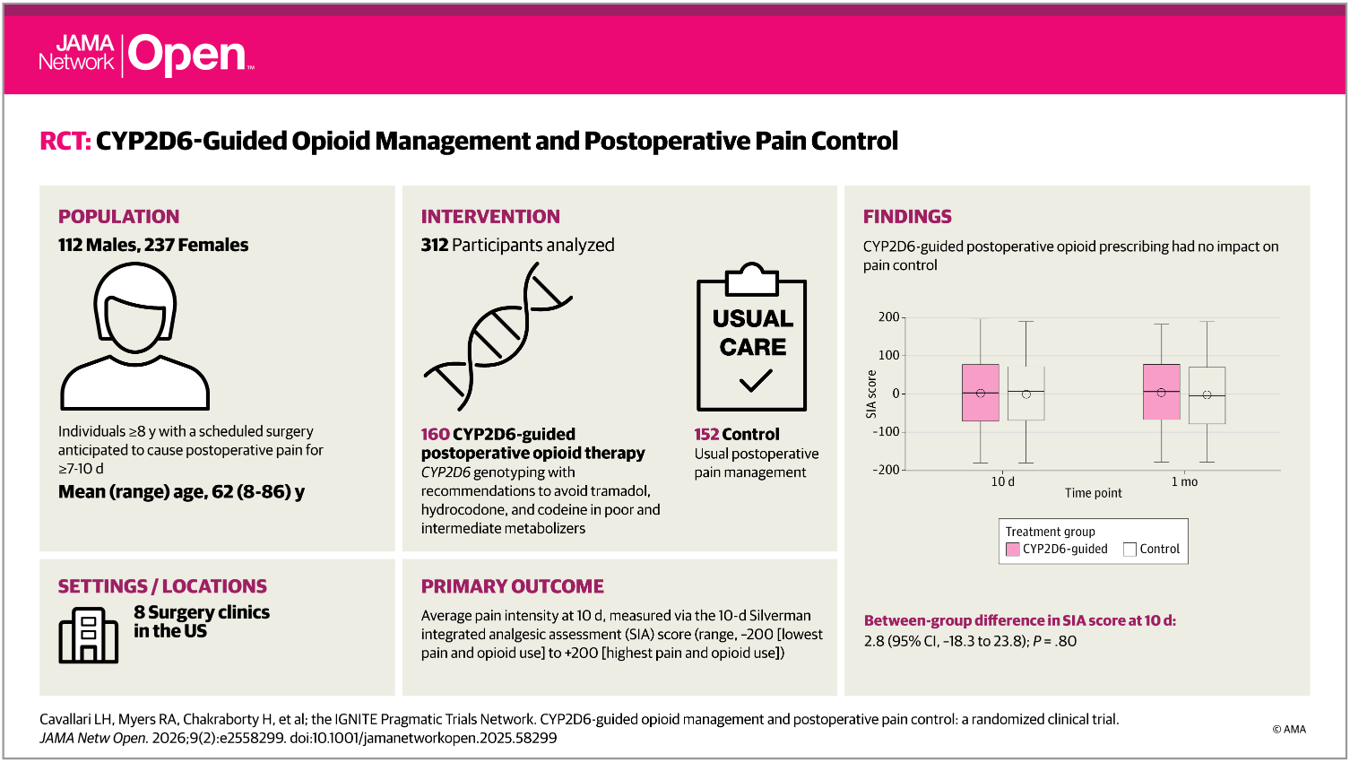
ADOPT-PGx Acute Pain Manuscript Published with Invited Commentary
The ADOPT-PGx study examined the impact of genotype-guided opioid prescribing on postoperative pain management. Results from the study were recently published in JAMA Network Open, accompanied by an invited commentary […]...
CYP2D6-Guided Opioid Management and Postoperative Pain Control
Larisa H. Cavallari, PharmD1; Rachel A. Myers, PhD; Hrishikesh Chakraborty, DrPH DOI: 10.1001/jamanetworkopen.2025.58299 Article on Jama Network...
MUSC researchers develop training for community health workers to improve cancer genetic testing
To address low participation in genetic testing, a team of researchers from the Medical University of South Carolina, led by Caitlin G. Allen, PhD, MPH, developed a 10-module training curriculum […]...
FDA approves safety labeling changes regarding DPD deficiency for fluorouracil injection products
On March 21, 2024, the Food and Drug Administration (FDA) approved safety labeling changes for fluorouracil injection products. Revisions to the labeling note increased warning of severe toxicity in patients […]...
GUARDD-US Team Surpasses Enrollment Goal
The GUARDD-US study aims to determine the impact of disclosing genetic of kidney failure among adults with African ancestry on blood pressure. The GUARDD-US study team celebrated surpassing their enrollment […]...
ADOPT PGx Team Reaches End-Of-Enrollment Milestone
ADOPT PGx recently celebrated reaching its participant enrollment milestone in September 2023 following the enrollment of 4,111 total participants. The last participant should complete the follow-up period for the trial […]...
CCPM’s Biobank unearths disease risk and pinpoints problems with medications
The biobank at the Colorado Center for Personalized Medicine (CCPM), a partnership between UC Health and the University of Colorado Anschutz Medical Campus, uses genetic data to identify specific genetic […]...
NIH funds new Genomics and Public Service Fellowship Program
The National Human Genome Research Institute (NHGRI), part of the National Institutes of Health, has awarded the American Society of Human Genetics (ASHG) a five-year, $7.1 million contract to support […]...
Scientists release a new human “pangenome” reference
Researchers have released a new high-quality collection of reference human genome sequences that captures substantially more diversity from different human populations than what was previously available. The work was led […]...
NASEM Report Says Researchers Need to Rethink and Justify How and Why Race, Ethnicity, and Ancestry Labels Are Used in Genetics and Genomics Research
Researchers and scientists who utilize genetic and genomic data should rethink and justify how and why they use race, ethnicity, and ancestry labels in their work, says a new National […]...
The University of Pittsburgh Enrolls First GUARDD-US Participant
Congratulations to the University of Pittsburgh! The site was activated on Monday, December 5 and enrolled their first participant on Wednesday, December 14, 2022....
PGx ECHO Program
The University of Minnesota College of Pharmacy invites all actively practicing clinicians to join their Pharmacogenomics (PGx) ECHO project. PGx ECHO aims to improve health professionals’ confidence in using PGx […]...
UAB Enrolls First GUARDD-US Participant
Congratulations to the University of Alabama at Birmingham (UAB)! The site was activated on Wednesday, September 14 and enrolled their first participant on Friday, September 16, 2022....
NIH Genetics/Genomics Competency Center is refreshed and renamed to GenomeEd
The National Human Genome Research Institute’s Genetics/Genomics Competency Center (G2C2) has been a valued resource for healthcare provider educators and learners alike for over 10 years. The resource, now called […]...
Cancer Centers Nudge Oncologists Toward DPYD Testing as PGx Supporters Push For Guidelines Change
Read the full article on GenomeWeb....
Implementing a Pragmatic Clinical Trial to Tailor Opioids for Acute Pain on behalf of the IGNITE ADOPT PGx Investigators
Larisa H Cavallari, Emily Cicali, Kristin Wiisanen, Roger B Fillingim, Hrishikesh Chakraborty, Rachel A Myers, Kathryn V Blake, Bolanle Asiyanbola, Jordan F Baye, Wesley H Bronson, Kelsey J Cook, Erica N Elwood, Chancellor F Gray, Yan...
WHO’s Science Council Launches Report Calling for Equitable Expansion of Genomics
The World Health Organization (WHO) Science Council of experts has issued its first report, on accelerating access to genomics for global health. The report argues that it is not justifiable […]...
NHGRI Concludes Genomics and the Media Seminar Series
In June, the National Human Genome Research Institute (NHGRI) concluded the seven-part virtual “Genomics and the Media Seminar Series.” The series featured trailblazers in science communication. Each speaker, an expert […]...
Medical Genomics in Underrepresented Populations from Latin America to the Pacific
On June 13, Dr. Andrés Moreno Estrada delivered a lecture for the Genomics and Health Disparities Lecture Series, co-sponsored by institutes in the National Institutes of Health (NIH) and the […]...
NHGRI Releases New and Improved Talking Glossary of Genomic and Genetic Terms
On National DNA Day (April 25) this year, the National Human Genome Research Institute (NHGRI) released a fully revamped version of its popular talking glossary, which included a new name: […]...
Researchers Generate the First Complete, Gapless Sequence of a Human Genome
Researchers Generate the First Complete, Gapless Sequence of a Human Genome Scientists have published the first complete, gapless sequence of a human genome, two decades after the Human Genome Project […]...
Publication Available on the Design and Rationale of GUARDD-US
Design and rationale of GUARDD-US: A pragmatic, randomized trial of genetic testing for APOL1 and pharmacogenomic predictors of antihypertensive efficacy in patients with hypertension Abstract Rationale and objective: APOL1 risk...
Submit Ideas for Opportunity to Speak at Virtual NHGRI Symposium
The National Human Genome Research Institute (NHGRI) invites you to submit your ideas for an opportunity to be one of six speakers at its upcoming virtual symposium, “Irreducible Subjects: Disability […]...
Call for Applications: Joint Postdoctoral Fellowship in Clinical Ethics and the ELSI of Precision Medicine
The University of North Carolina (UNC) Center for Genomics and Society (CGS) and the Center for Bioethics (C:B), together with the Program for Precision Medicine in Health Care (PPMH), announce a joint […]...
Lori Orlando to present during Public Health Genetics Week
Public Health Genetics Week will take place May 23-27, 2022 and virtual events are free and open to the public. Read More>>...
Notice of Special Interest: Development and Implementation of Clinical Informatics Tools to Enhance Patients’ Use of Genomic Information
Interested in developing clinical tools to enhance how patients use genomic information? Read more and apply>>...
IGNITE’s GUARDD pilot study determines effects of testing genetic risk for kidney failure
The Implementing GeNomics In practice (IGNITE) Pragmatic Clinical Trials Network’s GUARDD-US study has published their main outcomes paper of the pilot study, whose aim is to determine the effect of […]...
Could a Community-Based Approach to Genetic Testing Help African Americans Reduce Risks of Chronic Kidney Disease?
Mount Sinai, lead site for GUARDD-US, saw drops in blood pressure for hypertensive patients, supporting further trials and extensive community involvement Read More>>...
IGNITE celebrates Medical Genetics Awareness Week
Natasha Petry, PharmD, MPH, BCACP, Pharmacogenomics Clinical Pharmacist at Sanford Imagenetics is part of Vanderbilt University’s clinical site with the IGNITE Network. Learn More About ACMG>>...
UNC’s Joint Postdoctoral Fellowship in Clinical Ethics and the ELSI of Precision Medicine
IGNITE network affiliate, UNC Program for Precision Medicine in Health Care, has posted an opportunity for a joint postdoctoral position in clinical ethics & the ethical, legal, and social implications […]...
RFA: Supporting Talented Early Career Researchers in Genomics (R01 Clinical Trial Optional)
This Funding Opportunity Announcement is intended to identify and support research projects by exceptionally promising Early Stage Investigators with a long-term career interest in pursuing innovative research in genomics. This […]...
Notice of Special Interest: Development and Implementation of Clinical Informatics Tools to Enhance Patients’ Use of Genomic Information
The National Human Genome Research Institute has issued Notice of Special Interest NOT-HG-22-011 to encourage applications to develop and implement patient-facing genomic-based clinical informatics tools that facilitate or enhance...
CAGPM’s MeTree software recommended by President’s Cancer Panel
Lori Orlando (Duke Center for Applied Genomics & Precision Medicine) and the MeTree team highlighted in the Panel’s Closing Gaps in Cancer Screening Report to President Biden Read More>>...
New Leadership Announced for Coordinating Center
The Implementing GeNomics In practice (IGNITE) Pragmatic Clinical Trials Network is an NIH-funded network dedicated to supporting the implementation of genomics in healthcare. Effective January 2022, Christina Wyatt, MD, associate...
Opportunity to Publish, a special issue of the Journal of Personalized Medicine
A Special Issue of the Journal of Personalized Medicine will present cutting-edge research and commentary to characterize the future of personalized medicine. Papers from multiple disciplinary perspectives focus on topics that...
Multisite evaluation of institutional processes and implementation determinants for pharmacogenetic testing to guide antidepressant therapy
Abstract There is growing interest in utilizing pharmacogenetic (PGx) testing to guide antidepressant use, but there is lack of clarity on how to implement testing into clinical practice. We administered […]...
Why Knowing Your Family’s Health Past Benefits Your Future
Lori Orlando, MD, MHS, MMCI, Director of the Precision Medicine Program in the Center for Applied Genomics and Precision Medicine at Duke University, recently contributed to the Duke Health blog […]...
NHGRI Fact Sheets about Genomics
The National Human Genome Research Institute (NHGRI) has produced this series of fact sheets to explain complex concepts in genomics research to a non-scientific audience. Teachers, students and the general […]...
Journal of Personalized Medicine publishes papers from IGNITE Affiliate, Arkansas Children’s Hospital
Journal of Personalized Medicine recently published two papers featuring researchers from IGNITE Affiliate, Arkansas Children’s Hospital: Molecular Dysregulation in Autism Spectrum Disorder Implementing Pharmacogenomics Testing:...
Ginsburg named Chief Medical & Scientific Officer, NIH All of Us Research Program
Geoffrey Ginsburg, MD, PhD, a professor of Medicine (Cardiology), Pathology, Biostatistics and Bioinformatics, and Biomedical Engineering, and a professor in School of Nursing, will leave Duke to assume the role […]...
Call for Applications: NIH-ACMG Fellowship in Genomic Medicine Program Management
The National Institutes of Health (NIH), in partnership with the American College of Medical Genetics and Genomics (ACMG), seeks qualified physicians, physician assistants, nurse practitioners and genetic counselors interested in […]...
Lori Orlando to speak at WSU College of Medicine Genomics & Adoption Symposium
Lori Orlando, MD, MHS, MMCI, director of the precision medicine program in the Center for Applied Genomics & Precision Medicine at Duke University and IGNITE co-PI, will speak at the Washington State […]...
GUARDD-US Team Activates All Sites
Congratulations to the Genetic Testing to Understand and Address Renal Disease Disparities across the US (GUARDD-US) team for getting all sites activated approximately one month after reaching a milestone of […]...
IGNITE Diversity & Inclusion Statement
The IGNITE Network Leadership calls for Diversity and Inclusion in the conduct of research We call on scientific and medical communities to acknowledge and work toward eliminating the injustices perpetuated […]...
Fellowship in Genomic Medicine Program Management
The National Human Genome Research Institute (NHGRI) invites qualified physicians, physician assistants, nurse practitioners and genetic counselors to apply to the Fellowship in Genomic Medicine Program Management. This is an NIH...
UCSF TRANSPERS Center seeks Asst/Assoc. Prof of Health Services Research, Health Economics
UCSF is seeking a faculty member at the Assistant or Associate Professor rank with expertise in health services research, health economics, or a related field. Qualified applicants may also serve […]...
New Funding Opportunities for Genetic Counseling Research
NHGRI has two new funding opportunities available for genetic counseling research. Visit the NHGRI website for more information. Investigator-initiated research on genetic counseling processes and practices (R01, Clinical trial...
DCRI Think Tank Paper Looks to Future of Pragmatic Clinical Trials
Attendees to a January 2019 DCRI Think Tank outlined recommendations for optimizing pragmatic clinical trials and weighing risks and benefits of innovative trial designs. Read more from the Duke Clinical […]...
GUARDD-US Enrolls First Patient
IGNITE hit a major milestone on July 10, 2020: The University of Florida randomized the first patient in to the GUARDD-US pragmatic clinical trial. Genetic testing to Understand Renal Disease […]...
A Crystal Ball for the Decade Ahead
With a new decade dawning, Duke Today asked several Duke professors to hypothetically gaze into their crystal balls to tell us what they see happening in the years ahead. CAGPM […]...
Fellowship in Genomic Medicine Program Management
The National Human Genome Research Institute (NHGRI) invites qualified physicians, physician assistants, nurse practitioners and genetic counselors to apply to the Fellowship in Genomic Medicine Program Management. This is an NIH...
IGNITE PI Dr. Joshua Denny to serve as new Chief Executive Officer of All of Us research project
As CEO, Denny will oversee NIH’s efforts to build one of the largest and most comprehensive precision medicine research platforms in the world, in partnership with a diverse network of […]...
IGNITE blazes the way for genomic medicine
Getting diagnosed with a disease can be scary. Health providers work to find the right treatment plan based on the current standard of care guidelines. But what if in addition […]...
The Lancet publishes series of 5 papers featuring IGNITE researchers
Members of the IGNITE Network were involved in three of the five publications in this series. Genomic Medicine 1: Opportunities, resources and techniques for implementing genomics in clinical care describes the major […]...
Andrea Ramirez to present at the AMIA 2019 Annual Symposium
Dr. Andrea H. Ramirez will present her research, Extracting Drug Exposure Epochs and Drug Response Outcomes from Electronic Health Records, at the American Medical Informatics Association (AMIA) 2019 Annual Symposium on November […]...
Henry Ong to present at the AMIA 2019 Annual Symposium
Dr. Henry Ong will present his research, Extracting Drug Exposure Epochs and Drug Response Outcomes from Electronic Health Records, at the American Medical Informatics Association (AMIA) 2019 Annual Symposium on November 18. […]...
NIH funds clinical trials using genomics to treat chronic diseases
The National Institutes of Health will fund clinical trials to assess the benefits, applicability and efficacy of applying genomic medicine interventions to improve management of diseases such as high blood […]...
NHGRI publishes overview about IGNITE
The Implementing Genomic in Practice (IGNITE) Network was established in 2013 (RFA-HG-12-006, RFA-HG-12-007 and RFA-HG-13-004) to develop methods for incorporating genomic information into clinical care and explore the methods for...
PHASeR: A collaboration in PGx to help veterans
There are about 15 million cancer survivors in the United States; about 400,000 of them are also veterans in the Veterans Affairs (VA) system. As more people survive cancer, their […]...
Evaluating Genomic Medicine Interventions
The IGNITE I Network funded six genomic medicine projects. Through varied interventions, Lori Orlando, M.D., and team hypothesized that synergies across projects could be leveraged to better understand participants’ experiences […]...
Ginsburg presented at 1st Macrogen and LKCMedicine Scientific Conference
Geoff Ginsburg, M.D., Ph.D., presented at the one-day scientific conference “Applications of Genomic Advances in Healthcare” on March 18, 2019, in Singapore. His presentation was entitled, “Achieving Impact of Precision Medicine...


 GMKB
GMKB