Implementing GeNomics In pracTicE (IGNITE) Network, is an NIH-funded network dedicated to supporting the implementation of genomics in healthcare. The network started as IGNITE Genomic Demonstration Projects (IGNITE GDP) and later became IGNITE Pragmatic Trials Network (IGNITE PTN).
Launched in 2013, the initial phase, IGNITE GDP or IGNITE I, focused on a consortium of collaborative genomic medicine pilot projects aimed at demonstrating the feasibility of, and developing methods for, incorporating individual genomic findings into clinical practice.
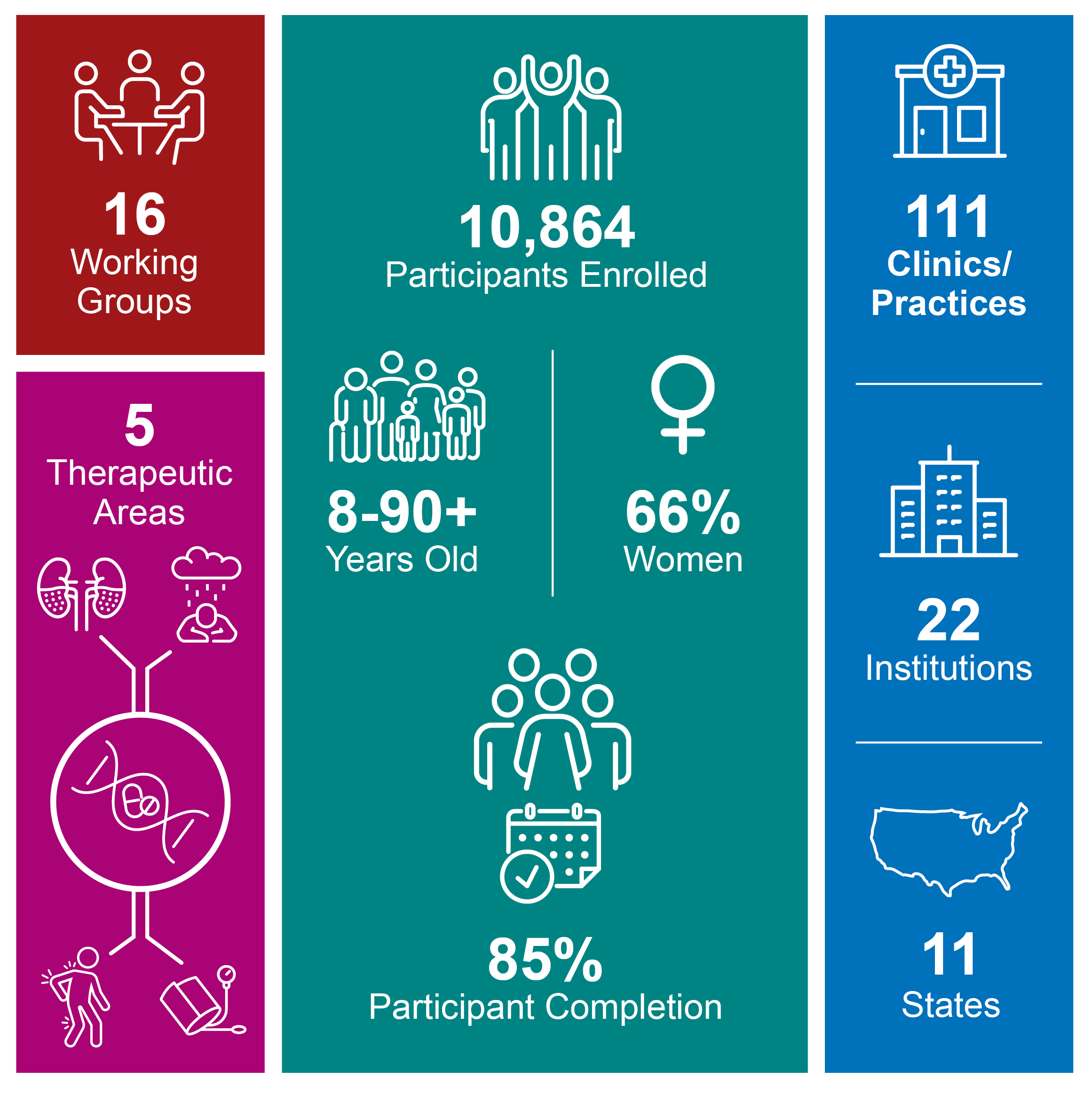
Building on the success of IGNITE I, the next phase of the network, IGNITE PTN or IGNITE II, began in 2018. This phase features five multi-site clinical groups and one coordinating center, that conduct pragmatic clinical trials of genomic medicine interventions across diverse settings and populations. The Protocol Review Committee (PRC), convened by the NHGRI, chose two studies – ADOPT PGx and GUARDD-US – based on their clinical importance, feasibility, potential impact, and cost.
The IGNITE website offers access to news, events, publications, the IGNITE Toolbox, implementation guides, and training opportunities.
![ADOPT-PGx Logo [no description + dash remov.]](https://gmkb.org/wp-content/uploads/2024/06/ADOPT-PGx-Logo-no-description-dash-remov-300x150.png) ADOPT PGx
ADOPT PGx
A Depression and Opioid Pragmatic Trial in Pharmacogenomics
Pain and depression are conditions that impact a substantial proportion of the U.S. population, but finding safe, effective drug therapies for both conditions is challenging. ADOPT PGx is a pragmatic clinical trial that enrolls patients into three pharmacogenomics (PGx)-guided therapy scenarios: acute post-surgical pain, chronic pain, and depression. For each scenario, participants will be randomized to genotype-guided drug therapy versus usual approaches to drug therapy selection ("usual care"). Changes in patient-reported outcomes representing pain and depression control using standard patient-reported outcomes measurement information system (PROMIS) scales define the primary endpoints. Secondary analyses include safety endpoints, changes in overall well-being, and economic impact represented by differences in healthcare utilization.
GUARDD-US
Genetic Testing to Understand and Address Renal Disease Disparities across the U.S.
GUARDD-US is a pragmatic clinical trial that aims to determine the effect of returning Apolipoprotein L1 (APOL1) genetic risk information to hypertensive African ancestry participants and their primary care providers on systolic blood pressure (SBP). The primary outcome is change in SBP from baseline to three months in participants with high-risk APOL1 variants (positives) comparing those who received results immediately versus those who received results at the end of the trial. APOL1 negative participants in the immediate arm were re-randomized to a genotype-guided approach to anti-hypertensive therapy versus usual care and three-month SBP were compared in both the immediate return of PGx results compared to the delayed return.
What is the IGNITE Toolbox?
The IGNITE Toolbox reflects a collection of expert consensus regarding special considerations, standard approaches, and best practices in the design, conduct, and reporting of genomic medicine. Given the rapid pace of change in this field, this Toolbox will continue to be added to and updated.



 GMKB
GMKB