Who are our Network Affiliates?
Our research network is comprised of members who believe in working together to provide evidence that implementing genomic science in clinical spaces will change the way we look at human health. Affiliate members are unfunded but committed individuals adding to the greater narrative around this goal.
What do affiliate members do?
- Contribute to evidence for genomic medicine by conducting studies and sharing findings with the network
- Support IGNITE trial project teams by doing supportive analysis, writing, etc.
- Survey the literature on a regular basis and add findings to the "Genomic Medicine Knowledge Base” (GMKB)
Affiliate members benefit from:
- Behind the scenes access to Network-developed tools and guidelines
- A team of scientists across many specialties who are actively engaged in the study of genomic medicine implementation
- A rich collaborative environment with Network Sites across the country
How do I apply to become an affiliate member?
We ask that investigators or organizations interested in becoming members submit an application via the button at the bottom of this page after reviewing the Affiliate Member Agreement.
Your application will include:
- A summary of rationale for participation including the goals and objectives of your initiatives
- The qualifications of your group (NIH biosketches may be used)
- A concise plan of network participation including:
-
-
- As appropriate, a specification of the genomic, phenotype, and implementation data that could be shared with the network
- The clinical implementation projects or areas that that will contribute to the goals of the IGNITE Network
-
-
- An assurance not to disclose confidential information obtained from other members
- An agreement to participate in IGNITE Network activities and abide by IGNITE policies as they apply to network activities
More information on applying to become a new member can be found under "New Affiliate Member Application Criteria"
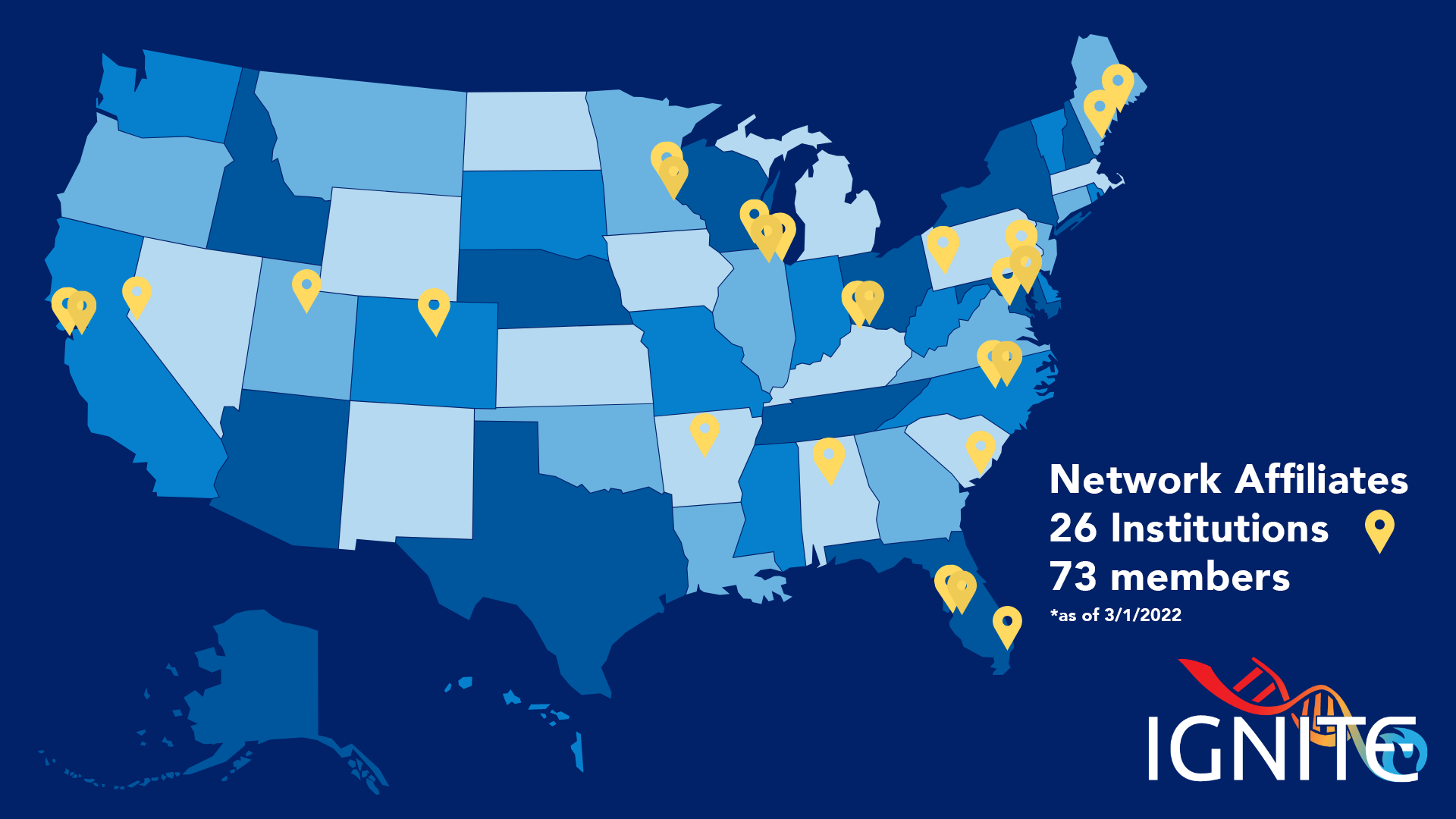
- Arkansas Children's Hospital
- Children's Minnesota
- Cincinnati Children's Hospital Medical Center
- Intermountain Healthcare
- Invitae
- Maine Medical Center
- MD Labs & Rxight Pharmacogenetics
- Medical University of South Carolina
- MedStar Health / Georgetown University Medical Center
- Moffitt Cancer Center
- Nicklaus Children's Hospital
- OneOme
- St. Elizabeth/University of Cincinnati
- Tempus Labs
- The Jackson Laboratory
- Uniformed Services University of the Health Sciences/Walter Reed National Military Medical Center
- University of Alabama at Birmingham
- University of California San Francisco
- University of Colorado
- University of Illinois at Chicago
- University of Illinois Cancer Center
- University of Michigan
- University of Minnesota
- University of North Carolina (UNC) Program for Precision Medicine in Health Care
- University of Pennsylvania Perelman School of Medicine
- University of Pittsburgh
Implementing Genomics In Practice (IGNITE) Genomic Medicine
Network Affiliate Collaborative Research Membership Agreement
Preamble
The Implementing GeNomics In pracTiCE Pragmatic Trials Network (IGNITE PTN, or “Network”) is an NIH-funded network dedicated to supporting the development of evidence for the applications of genomic medicine in healthcare. Several institutions within IGNITE PTN are implementing genomic medicine into clinical practice. Some of these groups are funded through an IGNITE PTN cooperative agreement, and the remainder are Network affiliates. There is an interest among these groups in working together to collect and share data relevant to genomic medicine evidence generation, inclusive of pharmacogenetics, risk assessment, complex disease management, and molecularly targeted therapies. To provide evidence that genomic medicine will improve patient outcomes, IGNITE PTN-funded members are conducting two network-wide genomic medicine pragmatic clinical trials (PCTs) – A Depression and Opioid Pragmatic Trial in Pharmacogenomics (ADOPT-PGx) and Genetic testing to Understand Renal Disease Disparities across the U.S. (GUARDD-US).
| ADOPT-PGx
This trial includes three pharmacogenomics (PGx)-guided therapy scenarios: acute post-surgical pain, chronic pain, and depression. For each scenario, participants are randomized to genotype-guided drug therapy versus usual approaches to drug therapy selection ("usual care"). Changes in patient-reported outcomes representing pain and depression control using standard patient-reported outcomes measurement information system (PROMIS) scales define the primary endpoints. Secondary analyses include safety endpoints, changes in overall well-being, and economic impact represented by differences in healthcare utilization. |
GUARDD-US
This trial that aims to determine the effect of returning apolipoprotein L1 (APOL1) genetic risk information to hypertensive African ancestry patients and their primary care providers on systolic blood pressure (SBP). The primary outcome is SBP at three months comparing patients with high-risk APOL1 variants (positives) versus no high-risk variants (negatives). The co-primary outcome is three-month SBP in positives versus those receiving delayed testing. Secondary outcomes include renal disease testing, and psycho-behavioral factors. APOL1 negative patients are randomized to a genotype-guided approach to anti-hypertensive therapy versus usual care with comparison of a three-month SBP. |
IGNITE PTN disseminates the methods and best practices its members develop in order to advance genomic implementation in healthcare. One outlet for public distribution is the IGNITE PTN Toolbox, which provides genomic medicine resources for clinicians and researchers. This toolbox is housed in the Genomic Medicine Knowledge Base (GMKB) where ideas can be shared so that everyone can begin to use this in practice, whether in community clinics or academic medical centers. IGNITE PTN scientists are interested in data dissemination, and so continue this collaboration in data aggregation with the assumption that de-identified aggregated data will be deposited into an IGNITE PTN database for use in future publications.
Collaborative Agreement
- IGNITE PTN “affiliate member” will be defined as an organization, institution or group of investigators within an institution participating in the effort. This AGREEMENT may be signed by one designate of the organization or institution. They will identify other participating affiliate members from their institution, and will ensure that all participants from their organization or institution abide by the principles set forth in this AGREEMENT. It is anticipated that participating affiliate members from one organization or institution may be involved in one or more specifically identified activities that are in alignment with their expertise and data contribution The IGNITE PTN anticipates not only a benefit from the affiliate member to further Network activities, as outlined below, but reciprocal benefit to the affiliate member through enhanced opportunities through participating in Network activities and attending Steering Committee meetings, engaging in collaborative studies with Network members, and authorship on Network publications.
- It is anticipated that there will be two categories of affiliate members within the IGNITE PTN, outside of the funded groups.
- ACADEMIC Affiliate Members. Academic affiliate members are those who are engaged in genomic medicine research and have data and/or knowledge and expertise to contribute to the collaboration. These members will have access to the compiled aggregate data, and may participate as contributing authors on resulting papers, as outlined in the Publications and Presentations Policies.
- INDUSTRY Affiliate Members. Industry affiliate members are employees of commercial organizations (pharma, device, third party payers, IT vendors, etc.) who are willing to contribute specialized technical or service function knowledge to the network. These members may participate in conference calls, face to face meetings and discussions, and serve as authors on resulting papers (with appropriate contributions for authorship) but may not have access to unpublished data.
Exceptions may be made for individuals who bring specific knowledge, expertise, and data (i.e. – from the conduct of genomics-guided clinical trials) deemed to be valuable to a resulting paper and/or network-sponsored clinical study. Designation as an Academic Affiliate member will be determined on a case by case basis by the Network Steering Committee, with a review of the statements of intent in the Affiliate member application specific to INDUSTRY collaborators
- In order to be a member of the IGNITE PTN, an individual or group affiliate member must agree to align with the objectives of the IGNITE PTN and engage in one or more of the following categories:
- Conducting studies (ancillary) that contribute to the evidence base for genomic medicine (not through the network’s PCTs, but through their own studies – eg trials, observational studies, etc.) and will share that evidence with the network, or
- Being part of an IGNITE PTN project team or working group and commit to doing work – analysis, paper/abstract writing – that benefits the PCTs, or
- Doing regular surveillance of the literature and provide the “Genomic Medicine Knowledge Base” (GMKB) with data/papers that contribute to the GMKBs mission of dissemination of genomic medicine knowledge and or contributing tool box artefacts and to keeping them updated.
- When relevant to ancillary studies (a study that uses some of the trial collected data plus other data, further defined by the IGNITE PTN Publications and Presentations (PPC) Policy affiliate members will designate points of contact for IGNITE Network staff to work with for each individual data set. These contacts should be very familiar with the details of the data sets, or have easy access to those who do.
- IGNITE PTN affiliate members with access to Network-generated data from the pragmatic trials being conducted agree that viewing these data is associated with responsibility for not publishing or disclosing it without approval of the IGNITE PTN Steering Committee.
- Affiliate members participating in ancillary studies with other network members agree to not publish or disclose shared data without approval of collaborating Network members, and abide by the IGNITE PTN PPC Policy.
- IGNITE PTN affiliate members are free to write papers based on their own data before, during, and after the activity’s association with this agreement.
- IGNITE PTN affiliate members may not write papers based on IGNITE PTN data without with explicit permission of those who collected the data. Members may suggest a manuscript idea to the Network that utilizes the compiled data and a decision will be made to move forward as a group, or submit a manuscript concept using Network pragmatic trial data to the Network Publications and Presentations Committee (PPC).
- All information and data disclosed during any Network-sponsored meetings are and shall remain the property of the disclosing institution. Neither the agreement nor any disclosure hereunder shall be deemed to vest in the receiving institution any license or ownership rights to the information and data or under any confidential information or inventions, patents, know-how, trade secrets, trademarks or copyrights owned or controlled by the disclosing institution or its affiliates.
- Access to the specific ancillary studies data sets and draft publications is limited to the collaborating members that have contributed data and members of the IGNITE PTN team providing support to ancillary studies. Those with major contributions to the respective cohorts will be expected to have lead roles in resulting manuscripts, and will have major roles (if they desire) in the analysis approach that is undertaken. Authors of IGNITE PTN ancillary studies publications will include all data contributors, data analysts, data curators, and writing group members who have participated in that specific ancillary study. Authors may be listed on the byline of the published paper or within appendices, as dictated by both contribution and the journal publishing the paper. It is expected that at least one author from each member group will be represented on the byline of the published paper as allowed by the publishing journal. The general rule for authorship order will be lead authors at the beginning and end, usually by level of contribution, and all other co-authors will be listed alphabetically in the middle. It is anticipated that there will be one designated leader of a specific project and the ultimate decision about author order at the beginning and end will reside with him/her, after consultation with these leading authors.
- With publication and release of the data on the public site, the IGNITE PTN will engage in appropriate dissemination, announcements, and emphasis of the availability of this exciting data set to the community.
- Investigators outside of the IGNITE PTN interested in access to data posted on public databases at the individual level will be required to apply for access. The members of the IGNITE PTN contributing data will establish the rules for accessing the individual level data by an outside investigator. If other data aggregation efforts arise from the IGNITE PTN, the IGNITE PTN members will similarly work to define the rules for access to those data. These data will be made available to other investigators after the publication of the relevant data.
Contribution Agreement
Our Institution agrees to participate in the activities in the following marked categories and adhere to the specific principles in this agreement:
- Conducting studies (ancillary) that contribute to the evidence base for genomic medicine (not through the network’s PCTs, but through their own studies – eg trials, observational studies, etc.) and will share that evidence with the network, or
- Being part of an IGNITE PTN project team or working group and commit to doing work – analysis, paper/abstract writing – that benefits the PCTs, or
- Doing regular surveillance of the literature and provide the “Genomic Medicine Knowledge Base” (GMKB) with data/papers that contribute to the GMKBs mission of dissemination of genomic medicine knowledge
IGNITE Network
Criteria for Affiliate Membership
The Implementing GeNomics In PracTicE (IGNITE) Network was established to enhance and accelerate theuse of genomic medicine by incorporating genomic information into clinical care and exploring methods foreffective implementation, diffusion, and sustainability. Its specific goals are to: (1) identify barriers and solutions to implementation of genomic medicine and disseminate best practices to the broader scientific community, and (2) determine the effectiveness of the use of genomic information in clinical care. IGNITE defines genomic medicine to include the use of genetic information and family history.
The IGNITE Network is open to all academic, government, and private sector scientists and organizations(including Industry) who are interested in participating in an open process to facilitate research in the application of genomic results to clinical care. An affiliate member can be an individual or an institutionwithin the categories listed above. Institutional affiliates should designate a principal investigator who willbe responsible for interacting directly with IGNITE leadership.
Affiliate members can benefit from:
- Access to Network-developed tools and guidelines, both published and beta versions
- Participation in specified Criteria for Participation described below with multidisciplinary scientistsactively engaged in the study of genomic medicine implementation
- A rich collaborative environment with Network Sites
Criteria for Participation in the IGNITE Network are:
- To align best with the objectives of the IGNITE network, affiliates would engage in one or more of the following categories:
- Conducting studies that contribute to the evidence base for genomic medicine (not through the network’s PCTs, but through their own studies – eg trials, observational studies, etc) and will share that evidence with the network, or
- Being part of an IGNITE II trial project team and commit to doing work – analysis, paper/abstract writing – that benefits the PCTs, or
- Doing regular surveillance of the literature and provide the “Genomic Medicine Knowledge Base” (GMKB) with data/papers that contribute to the GMKBs mission of dissemination of genomic medicine knowledge.
- Each participant will agree to share its clinical implementation strategies and/or experience with otherIGNITE sites for potential collaboration and adoption, and will collaborate and consider adoption of implementation measurements that are being developed in IGNITE.
- Participation in the Network should consist of substantial intellectual contributions to the Network, sharing of data as appropriate, and participating in network-wide research activities that will benefit both the network and the affiliate member.
- Each participant will adhere to Network policies, guidelines, and agreements as they pertain to network-related activities.
- Participants will take part in group activities, including attending Steering Committee meetings and working group calls, and contribute to the products of these group
- Each participant will agree that s/he will not disclose confidential information obtained from other members of the Network.
Additional criteria may be added upon recommendations of the IGNITE Steering Committee, External Scientific Panel, and NHGRI.
Affiliate Membership application and evaluation process:
An investigator or organization who is interested in applying for affiliate membership to the IGNITE Networkshould provide a description of no more than three pages of the contribution he or she proposes to make aspart of the Network, as well as a statement of agreement to abide by the Criteria for Participation listedabove, to the IGNITE Coordinating Center Staff.
Items that specifically should be addressed in the proposal and that will be used to evaluate acceptance into the Network are:
- A summary of the rationale for participation.
- The qualifications of the individual or, for institutional affiliates, the group of individuals designated askey personnel for that institution. NIH biosketches may be used and do not count against the 3 pagelimit.
- Clearly state the goals or objectives of your implementation initiative(s). This should include a concisedescription of the genomic medicine implementation projects and/or fields that the applicant leadsand/or that are ongoing at the institution in which the applicant has a leadership or active rol
- A concise plan of network participation, including:
- as appropriate, a specification of the genomic, phenotype, and implementation data that couldbe shared with the network, and
- the clinical implementation projects or areas that that will contribute to the goals of the IGNITENetwork.
- If appropriate: Evidence that the applicant’s projects are consistent with participants’ informed consent and the research has received appropriate IRB appro
- An assurance not to disclose confidential information obtained from other members of the Netw
- An agreement to participate in IGNITE Network activities and abide by IGNITE policies as they apply to network activities.
Evaluation criteria for affiliate membership will include:
- The applicant’s expertise and ability to participate fully in the Network
- The ability to share clinical implementation strategies and/or experience
- The ability to provide regular surveillance of the literature and contribute to the “Genomic Medicine Knowledge Base” (GMKB) with data/papers
- The ability to adopt implementation measurements that are being developed in IGNITE
- If applicable, the availability of genomic, phenotype, and implementation datasets and ability to sharethese data; and the diversity of the applicant’s subject population and ability to enrich the Network.
The participation of affiliate members will be assessed yearly by the Steering Committee.


 GMKB
GMKB