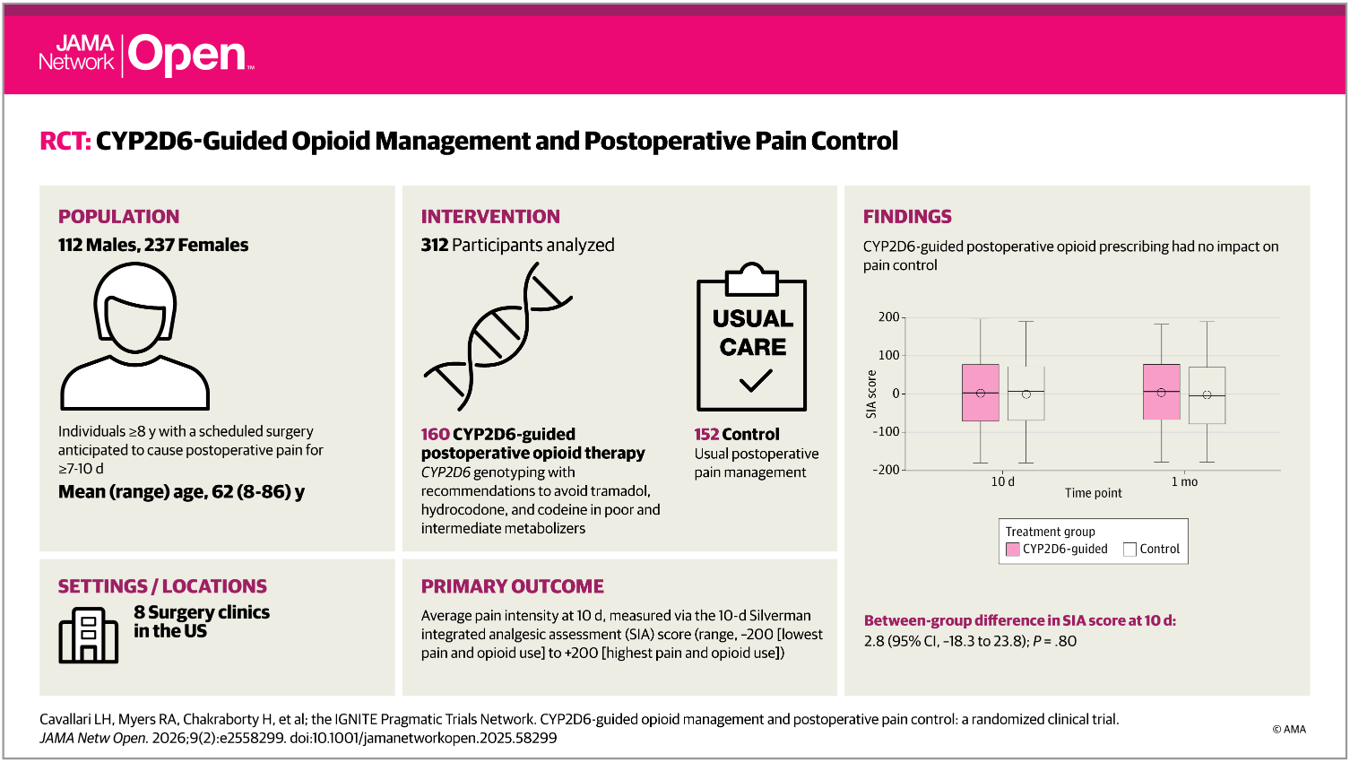
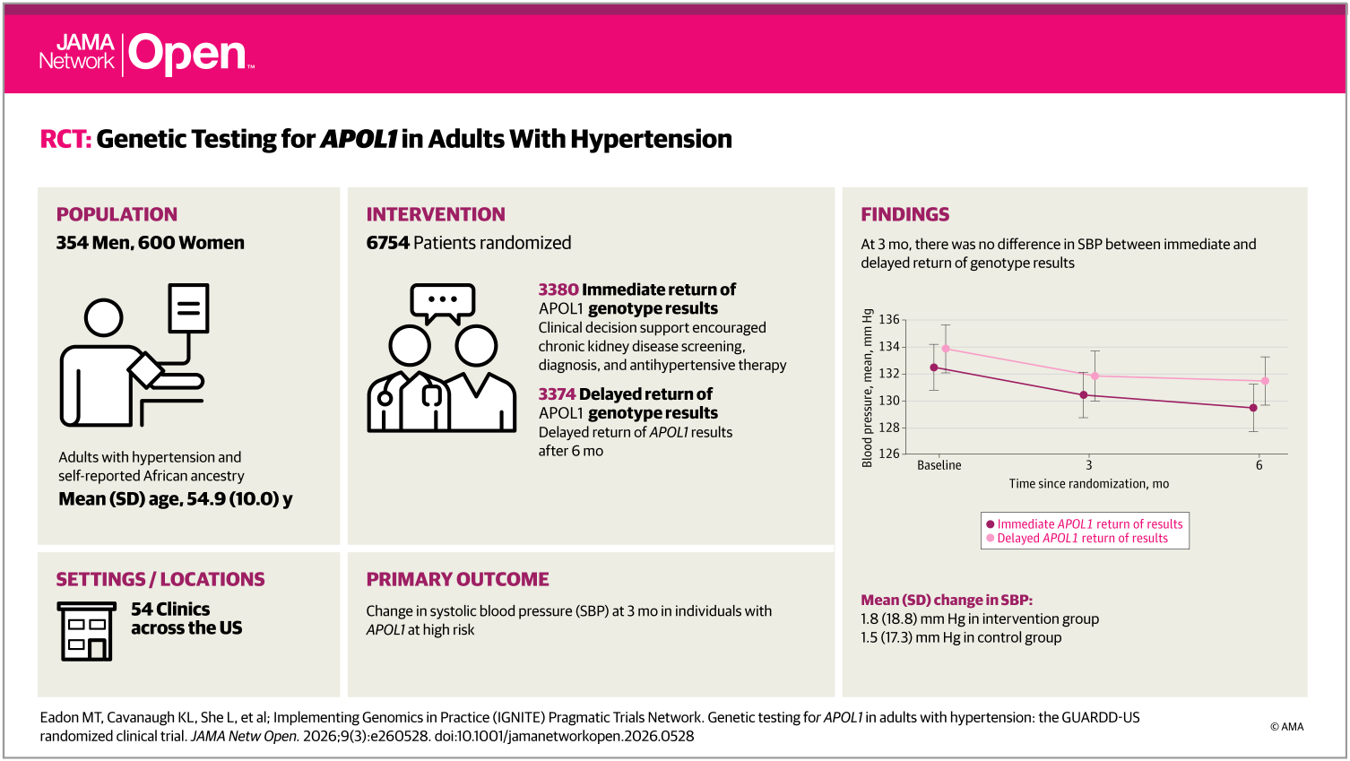
A new manuscript from the IGNITE Pragmatic Trials Network has been published in JAMA Network Open, describing the primary results of the GUARDD-US study. GUARDD-US is a randomized controlled trial examining the impact of returning APOL1 genetic risk results to patients with hypertension and their clinicians. The study explores whether knowledge of genetic risk for kidney disease can influence blood pressure management and chronic kidney disease (CKD) screening in routine clinical care. GUARDD-US enrolled more than 6,700 participants with self-reported African ancestry across 54 clinical sites in the United States, reflecting populations disproportionately affected by hypertension and CKD.
The primary outcome assessed changes in systolic blood pressure three months after randomization among individuals with APOL1 high-risk genotypes. Overall, immediate disclosure of genetic results was not associated with a significant reduction in systolic blood pressure compared with delayed disclosure. However, among participants who had uncontrolled blood pressure at baseline, those who received their genetic results experienced greater improvements in blood pressure control.
Importantly, returning APOL1 results also influenced clinical care beyond blood pressure management. Participants and clinicians who received immediate genetic results were more likely to pursue CKD screening and had higher rates of CKD diagnosis, highlighting the potential of genomic information to support earlier detection of kidney disease in at-risk populations.
Read the publication here.

 GMKB
GMKB